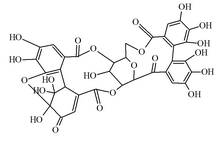
Granatin A
Appearance
| |
| Names | |
|---|---|
| IUPAC name
6,7,8,11,12,13,25,26,30,30,31,37-Dodecahydroxy-17,21,36,38,39-pentaoxaoctacyclo[18.16.1.12,19.127,31.04,9.010,15.023,28.029,34]nonatriaconta-4,6,8,10,12,14,23,25,27,33-decaene-3,16,22,32,3 5-pentone
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C34H24O22 | |
| Molar mass | 784.54 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Granatin A is an ellagitannin found in the pericarp of Punica granatum (pomegranate).[1] It is a weak carbonic anhydrase inhibitor.[1]
References
[edit]- ^ a b Satomi, H.; Umemura, K.; Ueno, A.; Hatano, T.; Okuda, T.; Noro, T. (1993). "Carbonic anhydrase inhibitors from the pericarps of Punica granatum L". Biological & Pharmaceutical Bulletin. 16 (8): 787–790. doi:10.1248/bpb.16.787. PMID 8220326.
